Gas Density in Diving: Research, Risks & Recommendations
Gas density is a proven risk factor in diving, yet many divers and instructors have never heard of it. So, to resolve that lack of education, I will explain the following critical points in this article:
- What gas density is, and how it affects divers
- The research studies leading our understanding
- The risks of high mix density at depth
- Recommended limits for the density of breathing mix
- How to calculate and apply the limits
Obviously, safety is paramount when diving. Make sure that you understand these issues and the recommended limits to keep yourself safe from the risks that come from them.
TL:DR – 5 Key Points on Gas Density in Diving
- The density of your breathing gas increases at depth.
- Research has shown that excessive density of breathing mixture has risks
- High gas density dramatically increases:
- Work of breathing (WOB)
- CO2 retention
- Elevated CO2 in the body causes:
- Rapidly debilitating CO2 narcosis
- Hypercapnia
- For these reasons, DAN has recommended gas density limits that significantly reduce risks. These equate to a maximum operating depth (MOD) for a given breathing gas mixture.
What is Gas Density?
Gas density, simply put, refers to the number of gas particles contained within a given volume.
In the context of diving, this concept holds immense significance as it directly influences the behavior of the gases that divers breathe. In turn, it influences respiratory efficiency, CO2 retention, narcosis, and overall safety.
Defining Gas Density in Diving
The density of a diver’s breathing mixture is influenced by two key factors: pressure and temperature.
As divers descend, the pressure around them increases. This causes gas molecules to be packed more closely together within the same volume. This heightened pressure results in an increase in gas density.
Likewise, temperature variations also impact the density of breathing gas. For instance, colder temperatures cause gas molecules to contract and increase density. Conversely, warmer temperatures have the opposite effect.
Research on Gas Density in Diving
Gas density, a once-overlooked factor in diving, has now taken the forefront of hyperbaric research, shedding light on its significant influence on divers’ safety and capabilities.
Recent groundbreaking work by Gavin Anthony and Simon Mitchell has provided profound insights into the role of the density of breathing mixtures in diving, reshaping our understanding of gas requirements and potential risks associated with improper gas density levels.
Study by Simon Mitchell and Gavin Anthony
In their paper titled “Respiratory Physiology of Rebreather Diving,” Anthony and Mitchell focused on both open-circuit and rebreather divers to reveal a pivotal connection between the density of breathing gas and various physiological responses.
Gas Density and CO2 Retention
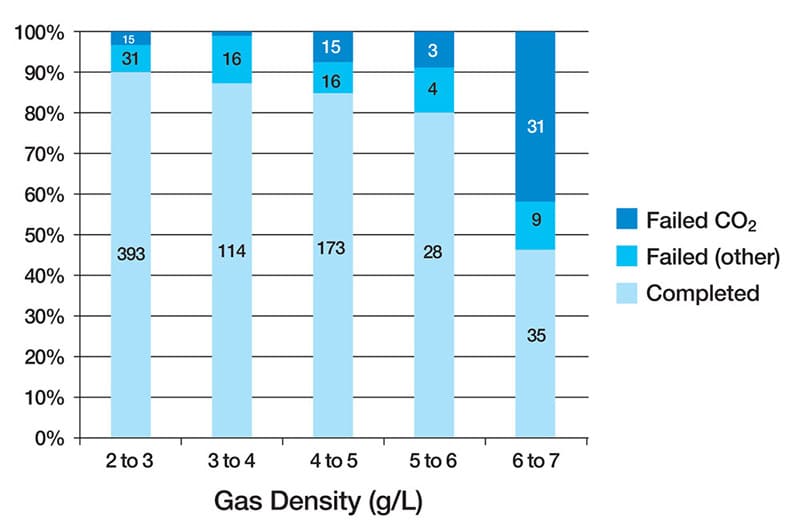
One striking finding of their research is that divers faced a critical risk of CO2 build-up when their respired gas density approached 6.0g/L.
This density threshold led to dangerous levels of CO2 retention, jeopardizing divers’ safety.
In earlier research, by Dan Warkander and colleagues, two participants experienced a state of CO2-induced incapacitation. Importantly, these individuals exhibited this incapacitation without being conscious of the symptoms they were experiencing.
Density, Depth, and Pressure
Diving deeper compounds the impact of gas density. For instance, the study indicated that at 4 ata (30m/100′), exhaled gas flow and ventilation rate dropped nearly in half compared to the conditions at 1 ATA.
Importantly, this depth-related effect further emphasized the necessity of managing gas density to ensure efficient respiration and gas exchange.
The Effect of Gas Density on Divers
Though imperceptible to our senses, the influence of gas density is substantial. The effects become more impactful as depth increases.
The density of breathing gas exerts its influence through two primary avenues:
- Work of breathing (WOB)
- Carbon dioxide (CO2) retention
In turn, the accumulation of CO2 in the body can cause:
- CO2 narcosis
- Hypercapnia
These interconnected factors play a pivotal role in divers’ physiological responses and safety beneath the waves.
Gas Density and Work of Breathing (WOB)
Gas density has a profound impact on divers’ work of breathing (WOB), which refers to the effort required to breathe while submerged.
An increase in the density of breathing gas intensifies WOB, necessitating greater energy expenditure for each breath.
- Elevated WOB leads to heightened CO2 production as divers use more energy for respiration.
- The ability to inhale fresh gas is compromised, amplifying the challenge of eliminating CO2.
- High WOB is believed to be a contributing factor to Immersion Pulmonary Edema (IPE).
- Factors like regulator performance, CCR functionality, respiratory rate, and even trim position also contribute to variations in WOB.
Gas Density and CO2 Narcosis
The interaction between gas density and CO2 elimination holds profound implications, particularly in the realm of CO2 narcosis.
Elevated CO2 levels can induce symptoms known as “dark narcosis,” marked by psychological unease that may escalate to active, uncontrollable panic.
- CO2 is an internal byproduct of respiratory metabolism and is 25 times more narcotic than nitrogen.
- Unlike nitrogen, CO2’s rapid onset contributes to its potent narcotic effects.
- CO2 narcosis can manifest as scuba diving anxiety, commonly referred to as “dark narc.”
- Novice divers are particularly susceptible to CO2 narcosis because a weaker skillset causes more exertion.
- Fast descents to depth can exacerbate CO2 narcosis
- Differentiating between CO2 narcosis and nitrogen narcosis is crucial for divers’ well-being and safety.
- Ascending to shallower depths can alleviate CO2 narcosis by reducing partial pressures.
Gas Density and Hypercapnia
Excess gas density can cause hypercapnia. This diving condition is characterized by excessive CO2 levels in the bloodstream due to compromised elimination. Understanding hypercapnia is paramount for maintaining a safe diving experience.
- Hypercapnia arises when the body’s respiratory mechanisms struggle to remove CO2 effectively.
- Hypercapnia symptoms range from shortness of breathweight to dizziness and headache.
- Severe cases of hypercapnia can lead to confusion, impaired cognition, and even unconsciousness.
- Gas density’s role in amplifying CO2 production underscores the importance of effective respiration strategies to prevent hypercapnia.
Recommended Gas Density Limits For Diving
DAN Recommended Limits
Divers Alert Network (DAN) has emerged as a prominent advocate for limiting the density of breathing mix, backed by the research findings from Anthony and Mitchell.
DAN’s recommendations establish clear gas density limits that align with specific diving depths. These guidelines ensure divers’ well-being and provide a framework for making informed gas choices.
Key points regarding DAN recommended breathing mix density limits include:
- An ideal gas density of 5.2 g/L is considered safe for diving.
- An absolute maximum limit of 6.2 g/L should be used.
- Using air, those limits calculate as:
- Ideal max depth 31m/102′
- Absolute max depth 39m/128′
- Commonplace dive planning using EANx/nitrox MOD calculation can exceed limits:
- The EANx32 MOD of 33m/110′ is 5.6 g/L
BSAC Gas Density Limits
The British Sub-Aqua Club (BSAC) has devoted substantial efforts to determining safe breathing mixture limits for diving. Through research spanning two decades, BSAC has established key recommendations that underscore the significance of adhering to a specific density of breathing gas thresholds for enhanced diving safety.
Key aspects of BSAC’s gas density limits are as follows:
- A recommended safe density of 5.2g/L or lower.
- Beyond 6.3g/L, divers face heightened risks of CO2 retention, oxygen toxicity, inert gas narcosis, decompression illness, and immersion pulmonary edema (IPE).
Specific depths with common gasses noted by BSAC include:
- Air at 5.1g/L = 30m
- Air at 6.3g/L = 40m
- 36% EANx at 5.2g/L = 30m
- 28% EANx at 6.4g/L = 40m
BSAC’s emphasis on gas density aligns with its commitment to diver safety, encouraging practitioners to make informed gas choices and plan dives that prioritize well-being.
Gas Density Limits and Technical END Limits
Technical diving training agencies like GUE and RAID recognize the importance of the density of breathing gas.
- Integration of Gas Density: GUE and RAID incorporate recommendations on breathing mix density into their training standards, employing trimix maximum Equivalent Narcotic Depth (END) limits to ensure diver safety.
- Maintaining Maximum END: Both GUE and RAID adhere to a maximum END of 30m/100′, to mitigate both inert gas narcosis and density of breathing gas concerns.
- RAID’s Recreational Deep Diver Option: RAID goes a step further by introducing helitrox/trimix as an option within their recreational Deep Diver training program, offering divers an additional tool to stay within recommended limits.
But I don’t have problems with CO2 on my dives!?
I am sure many divers will read this article who already have experience diving beyond the recommended density limits. In general, they won’t have observed significantly debilitating effects from CO2 retention and work of breathing. Understandably, it can be tempting to dismiss the dive safety advice.
However, do remember that respiratory demand increases enormously in an emergency. Under stress and physical workload, the addition of a high density of breathing gas can lead to rapid, catastrophic spikes in CO2 retention.
A sudden CO2 narcosis hit in the middle of dealing with a crisis underwater can make the difference between success and failure. As such, the density of your gas can be the factor that decides whether you survive, or not.
Calculating Gas Density in Dive Planning
The easiest way to calculate the density of breathing gas is to use this downloadable tool: simple gas density calculator from Brendon Allen.
The tool enables you to calculate your gas mix’s density at the maximum planned depth (in ATA). It also includes equivalent narcotic depth (END), and partial pressure of oxygen (PPO2) at the planned max depth.
Gas Densities at 1 ata
It is important to understand that the constituent gasses within diving mixes influence the overall density of your breathing gas.
| Gas at 1 ata | Density (g/L) |
|---|---|
| Helium | 0.179 |
| Nitrogen | 1.251 |
| Oxygen | 1.428 |
Gas Density Limits For Common Diving Mixes
The table below shows how gas density limits are easily exceeded if calculating dive maximum operating depth (MOD) solely from PPO2.
Gas | 1 ata Density | Ideal Max at 5.2g/L | Hard Max 6.2g/L | 1.4 PPo2 MOD |
|---|---|---|---|---|
Air | 1.293 | 30.4m | 38.1m | 56m |
EANx28 | 1.3 | 30m | 37.7m | 40m |
EANx32 | 1.31 | 29.8m | 37.4m | 33.7m |
EANx36 | 1.31 | 29.6m | 37.2m | 28m |
Gas Density in Diving Takeaways
In conclusion, you should try to incorporate considerations of gas density when planning and conducting your dives:
- On descend, the density of your breathing gas increases.
- Extensive research has highlighted the potential risks associated with excessive density of breathing gas, underlining the importance of understanding and managing this aspect.
A high density of breathing gas has been found to lead to:
- Increased work of breathing (WOB), potentially affecting divers’ performance and endurance.
- Enhanced CO2 retention, contributing to the risk of CO2 narcosis and hypercapnia.
Start applying the Divers Alert Network (DAN) recommended limits in your dive planning:
- Ideal limit – 5.2g/L
- Absolute limit – 6.2g/L
These limits correspond to a maximum operating depth (MOD) for specific breathing gas mixtures, offering divers a clear guideline for safer underwater exploration.
Ideally, use the gas density calculator, linked earlier in this article, to establish the deepest safe depth for your specific diving mix.
Finally, by acknowledging the risks of gas density and adhering to recommended guidelines, divers can minimize risks and ensure their underwater experiences remain both exhilarating and secure.
Essential Viewing: Simon Mitchell’s Lecture
This lecture by Simon Mitchell outlines the research done on gas density and how that led to limits being recommended.
References
- Anthony G, Mitchell S. Respiratory physiology of rebreather diving. In: Pollock NW, Sellers SH, Godfrey JM, eds. Rebreathers and Scientific Diving. Proceedings of NPS/NOAA/DAN/AAUS June 16-19, 2015, Workshop. Durham, NC; 2016; 66-79.
- Warkander DE, Norfleet WT, Nagasawa GK, Lundgren CEG. CO2 retention with minimal symptoms but severe dysfunction during wet simulated dives to 6.8 ATA abs. Undersea Biomed. Res. 1990; 17(6):515-523.
- Lambertsen CJ, Gelfand R, Lever MJ, Bodammer G, Takano N, Reed TA, et al. Respiration and gas exchange during a 14-day continuous exposure to 5.2% O2 in N2 at pressure equivalent to 100 FSW (4 ATA). Aerosp. Med. 1973; 44:844-849.
- Doolette DJ, Mitchell SJ. Hyperbaric conditions. Comprehensive Physiol. 2011; 1:163-201.
- Moon, R. E., Cherry, A. D., Stolp, B. W., & Camporesi, E. M. (2009). Pulmonary gas exchange in diving. Journal of Applied Physiology. https://doi.org/JAPPL-91104-2008
Further Reading
Performance Under Pressure, by Reilly Fogarty, DAN, 2019
Density Discords: Understanding and Applying Gas Density Research, by Reilly Fogarty, InDepth, 2019
Gas Density and Scuba, Diving Disease Research Center (DDRC), 2023
BSAC recommended diving gas mixtures for open circuit diving, BSAC website, 2020
About The Author

Andy Davis is a RAID, PADI TecRec, ANDI, BSAC, and SSI-qualified independent technical diving instructor who specializes in teaching sidemount, trimix, and advanced wreck diving courses.
Currently residing in Subic Bay, Philippines; he has amassed more than 10,000 open-circuit and CCR dives over three decades of challenging diving across the globe.
Andy has published numerous diving magazine articles and designed advanced certification courses for several dive training agencies, He regularly tests and reviews new dive gear for scuba equipment manufacturers. Andy is currently writing a series of advanced diving books and creating a range of tech diving clothing and accessories.
Prior to becoming a professional technical diving educator in 2006, Andy was a commissioned officer in the Royal Air Force and has served in Iraq, Afghanistan, Belize, and Cyprus.
In 2023, Andy was named in the “Who’s Who of Sidemount” list by GUE InDepth Magazine.
Purchase my exclusive diving ebooks!
Originally posted 2023-08-20 15:38:01.